October 2021 EQRS News

In This Issue:
Payment Year (PY) 2022 Preview Period
How to Navigate the ESRD Quality Incentive Program (QIP) User Interface (UI)
Calendar Year (CY) 2021 EQRS Data Submission Deadline
Download the PDF.
View past Newsletters.
Upcoming Events:
New User Training
- Tuesday, October 19, 2021 • 2:00-3:30 PM ET
Web Resources:
Conditions for Coverage for ESRD Facilities
EQRS Data Submission Update
On September 20, 2021, End Stage Renal Disease (ESRD) facilities began submitting the following January-September 2021 quality reporting data to the ESRD Quality Reporting System (EQRS):
- EQRS Clinical Data
- Clinical Depression Screening and Follow-Up Plan submissions
Additionally, all subsequent months in 2021 will open on the regular schedule for data submission beginning on the first day of each month (i.e., October opens October 1; November opens November 1; and December opens December 1). Calendar year (CY) 2021 In-Center Hemodialysis Consumer Assessment of Healthcare Providers and Systems (ICH CAHPS) Attestation will open for submission in EQRS on January 1, 2022.
The data submission deadline for all months in 2021 will be February 28, 2022, at 11:59 PM Pacific Time (PT). This data submission deadline applies to:
- EQRS Clinical Data
- Clinical Depression Screening
- ICH CAHPS Attestation
New EQRS Security Measure
Effective October 15, 2021, the Centers for Medicare and Medicaid Services (CMS) will launch an automated security measure that will disable the EQRS user role(s) for users that have not logged into their EQRS account within 60 days from their last login. EQRS users approaching the 60-day inactivity deadline will receive login inactivity alerts via email notification from eqrssupport-noreply@cms.hhs.gov at days 50, 53, 55, 57, and 59. Users must login to their EQRS account to reset this timer.
To reactivate their EQRS user role(s), users will need to login to EQRS using their Healthcare Quality Information System (HCQIS) Access Roles and Profile (HARP) account credentials. For step-by-step instructions on how to re-request your EQRS user role(s), refer to slides 36-45 of the HCQIS HARP Training available on MyCROWNWeb.
CMS strongly recommends those with EQRS user role(s) log into EQRS NO LATER THAN November 1, 2021, in preparation for the start of the Payment Year (PY) 2022 ESRD Quality Incentive Program (QIP) Preview Period. Following this recommendation will help to ensure necessary access during the ESRD QIP Preview Period. To login and verify your EQRS role(s) click here: https://eqrs.cms.gov/globalapp/.
PY 2022 ESRD QIP Preview Period
The PY 2022 ESRD QIP Preview Period will open in early November 2021 and remain open for 30 days. Communication will be provided as soon as the Preview Period date is determined.
During the PY 2022 ESRD QIP Preview Period, CMS recommends that organizations take the following steps:
- Facility or Corporate Points of Contact (POCs) and Viewers should review scores and reports via the ESRD QIP User Interface (UI) in EQRS.
- Facility and Corporate POCs should use the ESRD QIP UI in EQRS to submit inquiries regarding PY 2022 scores before the Preview Period concludes.
Reports are accessible based on roles and availability. Reports include the Performance Score Report (PSR) and the Patient List Report (PLR).
PY 2022 ESRD QIP Preview Period supporting materials will be available from the QualityNet website in early November 2021. Additional supporting materials include the Guide to the PY 2022 ESRD QIP PSR and the PY 2022 ESRD QIP UI Quick Start Guide. Visit the QualityNet website in November 2021 to download the PY 2022 supporting materials by clicking on “Reports” or “Resources”.
Note: Per the CY 2022 ESRD Prospective Payment System (PPS) Proposed Rule, CMS has proposed that no facility would receive a PY 2022 payment reduction due to the impact of EQRS operational issues and the COVID-19 Public Health Emergency (PHE). To find out if this proposal will be finalized, refer to the Federal Register website to check for the published CY 2022 PPS Final Rule that will be posted in November 2021.
How to Navigate the ESRD QIP UI
EQRS users must use EQRS to access the ESRD QIP interface. Refer to the PY 2022 ESRD QIP UI Quick Start Guide for assistance in navigating and/or obtaining access to the QIP application within EQRS. The QIP application allows Facility Viewers, Facility POCs, Corporate Viewers, and POCs to conduct the following activities within the QIP application:
- View QIP performance scores and feedback
- Submit, save, and reply to Preview Period inquiries (Facility/Corporate POC only)
- View previously submitted Preview Period inquires
- View and download Preview Period reports
Access the ESRD QIP application though the EQRS login page here: https://eqrs.cms.gov/globalapp/.
New Feature in EQRS
Since the re-opening of EQRS for clinical data submission in July 2021, CMS has been working on new features to enhance the data collected in EQRS. The following new EQRS feature is now available in EQRS:
Standard Kt/V Method:
Standard is a new Kt/V method in EQRS under the Manage Clinical page. This allows EQRS users to report Kt/V and the required ultrafiltration rate (UFR) data elements when the Standard Kt/V method is used.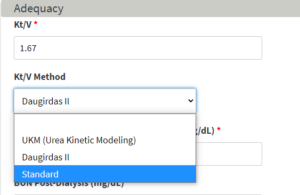
Please note that, per the ESRD QIP Measure Technical Specifications, facilities should select UKM or Daugirdas II methods for all patients receiving >2 and <4 treatments per week. This ensures that the Kt/V lab value is included in the Kt/V Dialysis Adequacy – Comprehensive measure calculation.
QualityNet Question and Answer Tool
Effective October 29, 2021, the CRAFT mailbox will no longer be monitored, and emails will not be answered. All EQRS Technical Assistance questions should be submitted to QualityNet Question and Answer (Q&A) Tool.
To submit a question through the Q&A Tool, select ESRD QIP from the Program drop-down list under Topic, and then select EQRS Technical Assistance from the General ESRD QIP sub-menu to submit your question.
When submitting your question(s) through the Q&A Tool, please be as detailed as possible to ensure your question and/or issue is effectively answered.
From the Chat Box
Question: A new dialysis patient just started with us last week and the nephrologist has declined to sign the CMS-2728 form because the patient is an acute kidney injury (AKI) patient. What should I do?
Answer: The nephrologist is correct to refuse to sign the CMS-2728. Facilities should not complete a CMS-2728 form for acute renal failure patients. Acute renal failure is a condition in which kidney function is expected to recover after a short period of dialysis (i.e. several weeks or months). Additionally, AKI patients should not be admitted in EQRS. Only ESRD patients are admitted into EQRS. If you have additional questions related to EQRS, please contact your local ESRD Network or the QualityNet Service Center via the Q&A Tool. in November 2021.
CY 2021 EQRS Data Submission Deadline
Dialysis facilities must submit all CY 2021 data by February 28, 2022 to meet CMS reporting requirements. The data submission deadline applies to all collection types (Hemodialysis and Peritoneal Dialysis) and to all submission methods. Please note that CMS strongly recommends that facilities complete large data submissions and audit batch submitted data prior to the February 28, 2022 data submission deadline.
If you batch submit or use the Health Information Exchange (HIE)/Electronic Data Interchange (EDI) methods, please follow the guidance provided by your HIE/EDI batch team within your organization.
CY 2021 data will support the PY 2023 ESRD QIP. For additional details, please refer to the CY 2022 ESRD PPS Proposed Rule, available from the Federal Register.
| EQRS Clinical Data Clinical Depression Screening and ICH CAHPS Attestation* |
|
| Reporting Month | 2020 and 2021 Data Submission Deadlines |
| September 2020 – December 2020 | September 15, 2021 at 11:59 PM PT |
| January 2021 Clinical Month | February 28, 2022 at 11:59 PM PT |
| February 2021 Clinical Month | |
| March 2021 Clinical Month | |
| April 2021 Clinical Month | |
| May 2021 Clinical Month | |
| June 2021 Clinical Month | |
| July 2021 Clinical Month | |
| August 2021 Clinical Month | |
| September 2021 Clinical Month | |
| October 2021 Clinical Month | |
| November 2021 Clinical Month | |
| December 2021 Clinical Month | |
*CY 2021 ICH CAHPS Attestation will open for submission in EQRS on January 1, 2022.
The information included as part of this newsletter is current as of the date of release.